LASER SOURCES FOR MULTIPHOTON MICROSCOPY
Section n. 1
Laser sources for Coherent Raman Scattering Microscopy
State of the art and new technologies
How to choose the best laser source for your SRS experiments
Written by Andrea Pruccoli (ESR10) and Eric Michele Fantuzzi (ESR7)
Since its discovery, laser technology has found broad applications in many different fields. As an example, the ready availability of various laser sources led to a renaissance of Raman spectroscopy where it is used to drive the scattering process. Apart from infrared absorption spectroscopy, Raman spectroscopy is the most important technique for vibrational spectroscopy. Depending on the specific Raman technique used and the type of sample under analysis, many different types of lasers can be considered for Raman experiment.
The excitation wavelengths used commonly range from near infrared to the visible and UV. The use of shorter wavelengths gives significantly stronger spontaneous Raman scattering, higher spatial resolution in spontaneous Raman microscopy and high sensitivity detectors can be used. Its main drawback is the increased probability for fluorescence background generation. Furthermore, when working with biological samples using short wavelength excitation, the penetration depth is reduced and the high energy of the excitation beam can damage the specimens, especially in imaging where the light is strongly focused. Thus, often a compromise is sought by using excitation lasers in the red or near infrared spectral range. Apart from the laser wavelength, other parameters, like the stability of the output, its spectral quality, and the polarization, are nowadays optimized to a level that concerning these points even very compact laser sources provide excellent properties. [1]
While for spontaneous Raman spectroscopy and microscopy, nearly exclusively continuous wave lasers are used, more advanced techniques like time-resolved and non-linear Raman techniques require pulsed lasers systems. Stimulated Raman Scattering (SRS) and Coherent Anti-Stokes (CARS) both require two synchronized excitation lasers outputs. In SRS the vibrational transition is resonantly excited by the lasers and the signal is then detected as either a gain of the Stokes beam intensity (SRG) or as a loss of the pump beam intensity, (SRL). Both measurements are highly affected by signal fluctuations and they require the modulation of one of the beams and subsequent demodulation usually done by lock-in detection to recover the signal from the noise. This means that the noise floor of the laser source is an important parameter. In CARS instead, the resonantly excited vibration is probed by a third beam that could either be the pump or another laser. The CARS signal thus is not influenced by laser fluctuations as much as the SRS signal but it can be affected by non-resonant background interference.
These techniques have found widespread interest in the biological community. They complement fluorescence microscopy since they allow label free imaging but require more advanced pulsed lasers systems. The different pulsed laser systems used in CARS and SRS microscopy can be distinguished based on the nature of the active medium. Initially many pulsed lasers used dye solutions. These require periodic care and involve the handling of potentially hazardous chemicals. For the inconvenience of operation and high maintenance required for the systems, they are today mostly replaced by solid state bulk lasers and more recently by fiber lasers.
Mode-locked fiber lasers and solid state bulk lasers that pump optical parametric oscillators are widely used and are commercially available as turn-key systems like the picoEmerald from APE, shown in Fig.1. This fiber based OPO system can be directly coupled with the most common confocal microscopes and be used for SRS and CARS as it emits a fixed Stokes and a tunable pump beam which are already overlapped in space and time. [2]
This system can be easily coupled, for example, with a multi photon microscope, e.g. the SP8 system from Leica microsystems [3], which results in an instrument capable of multimodal imaging that can provide contrast from SRS, CARS, second harmonic generation (SHG) and fluorescence. This setup offers a robust and versatile working environment (Fig.2).


OPO systems provide maximum flexibility and can be used for more advanced techniques, like wide field microscopy. Wide field microscopy is a technique that has been gaining more and more attention in the last 15 years as demonstrated by the several examples found in literature [4-11].
Wide-field microscopy is a technique that competes with single-point scanning microscopy, in Fig.3 is shown the main difference between those two approaches. In wide-field, instead of focusing the laser at a point to activate a non-linear optical process, e.g. CARS, a large sample region is illuminated at once. Since the laser is no longer focused in a single point, but its power is dispersed over a wide region of space, it is obvious that the laser source used for this technique must be more powerful than a point scanning one. For this reason, it requires a mode locked laser coupled with an OPO.
One of the first experimental setups [4] inherent to this technique used a 1,064-nm mode-locked Nd:vanadate laser from High-Q Laser (Hohenems, Austria) with 10-W output power, which delivers a stable 7-ps, 76-MHz pulse train, while 10% of the output power is used as the CARS Stokes beam. The remaining 9 W is used for synchronously pumping the Levante optical parametric oscillator (OPO) from APE (Berlin, Germany) to generate the CARS pump beam. The Levante allows a wavelength tuning over four separate intervals covering the 1,560- to 1,860-nm range. The OPO signal is intracavity doubled to produce tunable radiation between 780 and 930 nm with output powers of ≈1.5 W at 76 MHz. The narrow bandwidth pulses (≈3.5 cm-1) with durations of 5 ps assure a sufficient spectral resolution and a high signal-to-background ratio. With this configuration it is possible to obtain a field of view (FOV) approximately equal to 300×300µm with a penetration depth of 125µm.
A more recent experimental setup for wide-field microscopy has been reported by Shen and co. in 2018 [5]. They used a 10W picosecond Nd:YVO4 laser by Attodyne (Toronto, Canada), with 1 MHz repetition rate and 10μJ pulse energy at 1064 nm. The beam has been split into two, one sent to a supercontinuum laser source by NKT Photonics (Birkerød, Denmark) and used as the Stokes beam, while the second sent to a delay stage and used as the pump beam. With this configuration they reached a FOV of approximately 50×50µm.
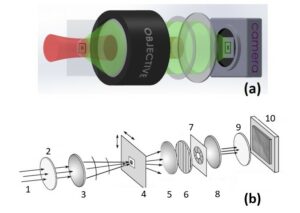
Fig.3 Comparison between wide-field configuration (a) and single-point scanning configuration (b). In (a), the excitation beam in red illuminates a wide area at the sample plane (black rectangle), while the CARS signal in green from the black cross is collected by an objective lens and imaged onto a camera. In (b), the laser beam scans over many regions of the sample and the scattered intensity pattern is recorded at each probe position. From left to right we have: 1 – Incoming laser beam; 2 –λ/2-wave plate; 3 – condenser lens; 4 – Sample holder; 5 – collecting lens; 6 – polarizator; 7 – filtering iris at image plane; 8 – imaging objective; 9 – narrow bandwidth spectral filter; 10 – CCD camera.
One of the current challenges of the wide-field microscopy community is enlarge the FOV of the sample up to mm2 or cm2 areas. Of course, get such CARS non-linear effect on a big area requires a lot of power, which require novel laser design and construction. Nowadays, thanks to the latest advances in laser sciences, is possible to design a high power femtosecond Yb laser pumping an optical parametric amplifier (OPA) to provide the pump and Stokes beams required to activate the CARS contrast over large (mm2) field of views. Both APE [12] and Light Conversion [13] (Vilnius, Lithuania), are part of the MUSIQ consortium and developed such technologies allowing to carry out wide-field experiments.
Examples of those devices are the AVUS Optical Parametric Amplifier by APE and the ORPHEUS Optical Parametric Amplifier by Light Conversion (LC), Fig.4. The first one is design to be coupled with a 1µm femtosecond laser and it is able to provide up to 50W of pump power (1MHz repetition rate). There are two different available versions of this OPA, < 200 fs and < 70 fs, both widely tunable in terms of wavelength, air cooled, and providing long-term temperature stability, completely automated via a specific software. On the other hand, LC is providing a similar system including a PHAROS high energy femtosecond laser that allows to reach powers close to 80W, pumping the ORPHEUS OPA provides a tunable output beam over a broad wavelength range. Also in the ORPHEUS case, it is possible to find several versions in terms of pulse duration and output energy.

While these OPO coupled systems provide maximum flexibility thanks to a wide tuning range and substantial power output, their complexity limits their usefulness when small and reliable closed box instruments are required (e.g. in clinical applications). In these cases, fiber lasers can offer some advantages over OPO systems. In a fiber laser the gain medium is an optical fiber usually doped with rare earth ions (Er3+, Nd3+, Yb3+, Pr3+, …) and the pumping is achieved by a fiber-coupled laser diode. The entire laser system can be contained in one housing with an automatic alignment. Furthermore, fiber laser based systems are usually cheaper than OPO coupled systems. The advantages of having a fiber laser based system has been demonstrated multiple times. Orringer employed it in 2017 in a portable instrument for intraoperative histopathologic diagnosis based on SRS, Fig. 5, that does not require staining and can be directly used in the operating room, reducing the operation time while maintaining or increasing the precision. [14] This setup is now commercially available by Invenio Imaging Inc. [15] Fiber lasers were also used in 2018 by Zirak et al. who implemented CARS for endoscopic contrast generation allowing molecular specific imaging of unlabelled samples. [16] In both applications, fiber lasers offer the advantage of being more robust and more compact than bulky OPO based laser systems with the drawbacks of having lower power performance and higher noise.
Either system provides advantages in their respective fields of use and can be employed for spectral imaging. OPO based systems are more versatile but, for many applications, the advantages are not worth the extra size and complexity. In these cases, fiber systems can be a cheaper and handier option.

Section n. 2
Light sources for harmonic generation and multiphoton fluorescence microscopy
Written by Thomas Deckert (ESR11)
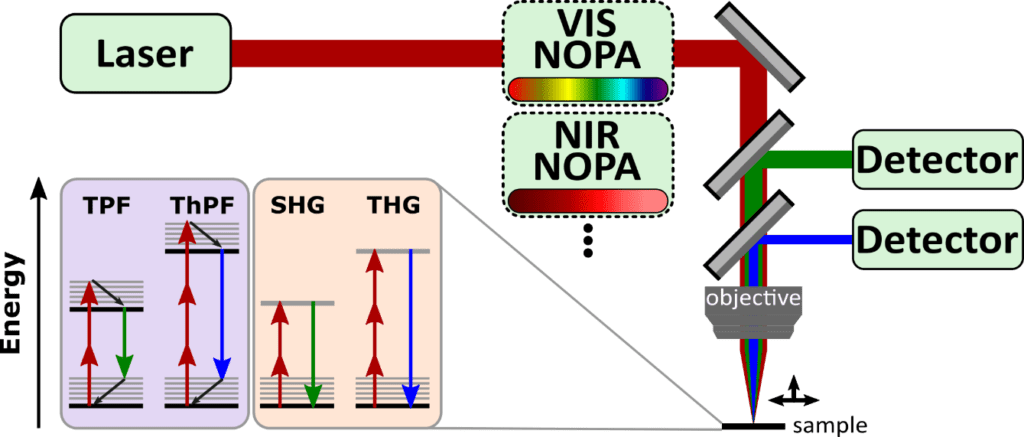
For many biophotonics applications, microscopy is an important technique for imaging biological specimen ex or in vivo. Confocal microscopy based on one-photon fluorescence therein is the simplest implementation, but limited in resolution, contrast, and signal quality. This is overcome by multiphoton microscopy (MPM), where imaging is based on nonlinear χ(2)-processes and beyond (e.g. two-photon and three-photon fluorescence (TPF, ThPF), second and third harmonic generation (SHG, THG)), and combines several advantages: higher resolution arising from a reduction of the effective size of excitation point spread function, 3D imaging, increased signal-to-noise ratio, less photodamaging, and label-free with endogenous fluorophores. Figure 6 shows an MPM setup in backward detection geometry as well as exemplary nonlinear processes in a simple sketch. Either the sample or the focussed light beam is moved in three-dimensional space to create an image of the object, which is exemplarily shown in Figure 7c-d. Undoubtedly the laser source is the most important ingredient in an MPM setup, and specific requirements on the source as well as current state-of-the-art technology is presented in the following paragraphs.
The underlying nonlinear processes in MPM experiments impose specific requirements on the laser sources used for imaging, which result in formulating an ideal laser source and is based on figures of merit proposed by C. Lefort [17][18]. Firstly, and most intuitively, pulsed radiation is favourable to drive nonlinear processes because it can achieve higher peak powers than continuous-wave radiation, and pulsed femtosecond lasers are superior to pico- or nanosecond laser sources. Consequently, and secondly, high average laser powers are desirable to increase the peak power. Thirdly, decreasing the repetition rate for constant average power will have a similar outcome, yet there is a trade-off that needs to be considered: For live imaging, a picture frame rate of at least 16 frames per second (fps) is desired, and higher repetition rates can push the fps or increase the image quality at constant frame rate. Fourthly, a broadband and tuneable excitation spectrum in and around the near-infrared will allow to simultaneously excite and detect several fluorophores, as exemplary depicted in Figure 6. This is achieved either by one or multiple laser sources directly, or by modifying the laser output with noncollinear parametric optical amplifiers (NOPAs) operating at different spectral windows to match the absorption spectrum of the desired nonlinear processes that are responsible for imaging.
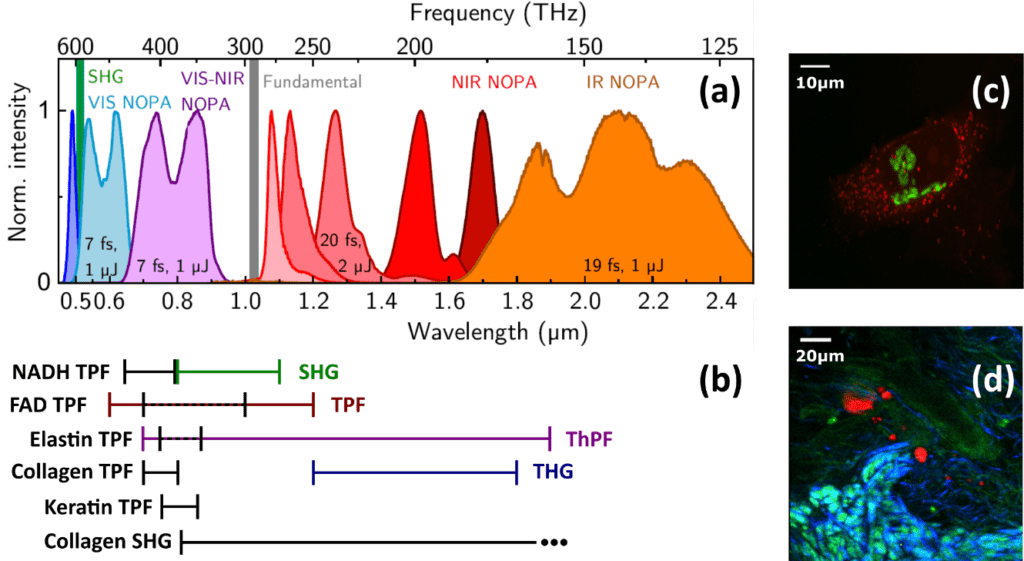
Altogether, an ideal laser source can be defined with the following parameters: 10 W average power (7 mW/nm), 2 MHz repetition rate, 100 fs pulse duration, and covering a spectral range between 500 nm and 1.9 µm [17]. When these parameters were formulated, Oshima et al. tested a Ytterbium (Yb) fiber laser that outperformed all others on the market at this time, solely lacking in high repetition rates [21]. Only recently, Grupp et al. reported on a regeneratively amplified 10 W Yb:KGW laser (PHAROS 10W, Light Conversion Ltd) that drives multiple noncollinear optical parametric amplifiers (NOPAs), producing broadband few-fs pulses with central wavelengths spanning from the visible to the infrared. It operates at 50 kHz repetition rate [19] and is easily adjustable up to 1 MHz. Figure 7a shows the accessible spectra of an expanded setup and demonstrates the excellent overlap with exemplary fluorophores (Figure 7b, black, left side) as well as the spectral windows required for most of the nonlinear processes (Figure 7b, coloured, right side). It closely matches the ideal parameters, and Light Conversion now offers a newer version of the PHAROS laser with higher repetition rates at this point in time. Most significantly however, this work demonstrates that optical parametric amplifiers (OPAs) and optical parametric chirped pulse amplifiers (OPCPAs) as well as fiber laser systems have tremendous potential to match the ideal parameters, especially the broadly tuneable spectral range and short pulse duration, whereas the average power and repetition rate are still subject to the laser that drives the amplifiers. They can either be custom-built, relying on an excellent review by Manzoni et al. [22], or are commercially available from many manufacturers, including APE, Fluence, Class 5 Photonics, Fastlite, EKSPLA, and Light Conversion. This year, Rico-Jimenez et al. reported on a broadband excitation source between 790 and 940 nm generated via a photonic crystal fiber based on a 80 MHz laser system with that provides 100 fs output pulses [23].
Concluding, the development of new laser sources at the edge of technology is a key factor to increase the quality of multiphoton microscopy in terms of resolution, data quality, imaging speed, and simultaneous imaging of multiple fluorophores. Nowadays laser sources continuously approach the proposed ideal parameters both in industry and research. Instead of utilizing the laser source directly for excitation, customly modifying the laser output to match individual requirements through nonlinear processes becomes increasingly present in current research, and industry also picked up on this challenge to provide versatile excitation sources, providing a prosperous future for multiphoton microscopy.
About the Authors
The MUSIQ Innovation Newletter n.1 was written by three of the Early Stage Researchers (ESRs) working on individual projects as part of the MUSIQ project.

Eric Michele Fantuzzi (ESR7)
Eric ist hosted at the Institut Fresnel at Centre National de la Recherche Scientifique developing an ultra-sensitive background free microscopy scheme for vibrational imaging by means of stimulated Raman scattering (SRS).

Andrea Pruccoli (ESR10)
Andrea is hosted at the University of Konstanz in the Chemisty Department and the goal of his research project is to do non-linear Raman microscopy of biomolecules with high sensitivity and will I will develop an experimental setup suited for CARS and SRS imaging studies with electronic pre-resonance enhancement.

Thomas Deckert (ESR11)
Thomas is hosted at Physics and Materials Science Research Unit at the University of Luxembourg his research targets the development of an experimental setup that will combine the generation of ultrabroadband pulses in the mid-IR spectral region with their coherent superimposition for multidimensional spectroscopy.
References
[1] Karlsson, H. and Illy. E. 2018, August 1. How to choose a Laser. Retrieved from https://www.laserfocusworld.com/lasers-sources/article/16555207/how-to-choose-a-laser-how-to-choose-a-laser-for-raman-spectroscopy
[2] APE. 2020, March 6. Picosecond Laser Widely Tunable. Retrieved from https://www.ape-berlin.de/en/picosecond-laser-tunable/
[3] Leica microsystems. 2020, July 15. Deep In Vivo Explorer SP8 DIVE. Retrieved from https://www.leica-microsystems.com/products/confocal-microscopes/p/dive/
[4] Evans, C. L., Potma, E. O., Puoris’haag, M., Côté, D., Lin, C. P., & Xie, X. S. (2005). Chemical imaging of tissue in vivo with video-rate coherent anti-Strokes Raman scattering microscopy. Proceedings of the National Academy of Sciences of the United States of America, 102(46), 16807–16812. https://doi.org/10.1073/pnas.0508282102
[5] Shen, Y., Wang, J., Wang, K., Sokolov, A. V., & Scully, M. O. (2018). Wide-field coherent anti-Stokes Raman scattering microscopy based on picosecond supercontinuum source. APL Photonics, 3(11). https://doi.org/10.1063/1.5045575
[6] C. Heinrich, A. Hofer, A. Ritsch, C. Ciardi, S. Bernet, and M. Ritsch-Marte, “Selective imaging of saturated and unsaturated lipids by wide-field CARS microscopy,” Opt. Express 16, 2699–2708 (2008). https://doi.org/10.1364/oe.16.002699
[7] Toytman, I., Cohn, K., Smith, T., Simanovskii, D., & Palanker, D. (2007). Wide-field coherent anti-Stokes Raman scattering microscopy with non-phase-matching illumination. Optics Letters, 32(13), 1941. https://doi.org/10.1364/ol.32.001941
[8] Toytman, I., Simanovskii, D., & Palanker, D. (2009). On illumination schemes for wide-field CARS microscopy. Optics Express, 17(9), 7339. https://doi.org/10.1364/oe.17.007339
[9] Lei, M. (2011). Video-rate wide-field coherent anti-Stokes Raman scattering microscopy with collinear nonphase-matching illumination. Journal of Biomedical Optics, 16(2), 021102. https://doi.org/10.1117/1.3533707
[10] Silve, A., Dorval, N., Schmid, T., Mir, L. M., & Attal-Tretout, B. (2012). A wide-field arrangement for single-shot CARS imaging of living cells. In Journal of Raman Spectroscopy (Vol. 43, Issue 5, pp. 644–650). https://doi.org/10.1002/jrs.4051
[11] Heinrich, C., Bernet, S., & Ritsch-Marte, M. (2004). Wide-field coherent anti-Stokes Raman scattering microscopy. Applied Physics Letters, 84(5), 816–818. https://doi.org/10.1063/1.1641164
[12] https://www.ape-berlin.de/de/
[13] http://lightcon.com/
[14] Orringer, D., Pandian, B., Niknafs, Y. et al. Rapid intraoperative histology of unprocessed surgical specimens via fibre-laser-based stimulated Raman scattering microscopy. Nat Biomed Eng 1, 0027 (2017). https://doi.org/10.1038/s41551-016-0027
[15] Invenio. 2020, September 5. High-Speed Label-Free Chemical Imaging. Retrieved from https://www.invenio-imaging.com/technology
[16] Zirak, P. et al. A rigid coherent anti-stokes Raman scattering endoscope with high resolution and a large feld of view. APL Photon. 3, 092409 (2018)
[17] Lefort, Claire (2017): A review of biomedical multiphoton microscopy and its laser sources. In Journal of Physics D: Applied Physics 50 (42), p. 423001. DOI: 10.1088/1361-6463/aa8050
[18] Lefort, Claire (Ed.) (2018): Laser sources in multiphoton microscopy: overview and optimization. Proc.SPIE (10677)
[19] Grupp, Alexander; Budweg, Arne; Fischer, Marco P.; Allerbeck, Jonas; Soavi, Giancarlo; Leitenstorfer, Alfred; Brida, Daniele (2017): Broadly tunable ultrafast pump-probe system operating at multi-kHz repetition rate. In Journal of Optics 20 (1), p. 14005. DOI: 10.1088/2040-8986/aa9b07
[20] Pope, Iestyn; Langbein, Wolfgang; Watson, Peter; Borri, Paola (2013): Simultaneous hyperspectral differential-CARS, TPF and SHG microscopy with a single 5 fs Ti:Sa laser. In Opt. Express 21 (6), pp. 7096–7106. DOI: 10.1364/OE.21.007096
[21] Oshima, Yusuke; Horiuch, Hideki; Honkura, Naoki; Hikita, Atsuhiko; Ogata, Tadanori; Miura, Hiromasa; Imamura, Takeshi (2014): Intravital multiphoton fluorescence imaging and optical manipulation of spinal cord in mice, using a compact fiber laser system. In Lasers Surg. Med. 46 (7), pp. 563–572. DOI: 10.1002/lsm.22266
[22] Manzoni, C.; Cerullo, G. (2016): Design criteria for ultrafast optical parametric amplifiers. In Journal of Optics 18 (10), p. 103501. DOI: 10.1088/2040-8978/18/10/103501
[23] Rico-Jimenez, Jose; Lee, Jang Hyuk; Alex, Aneesh; Musaad, Salma; Chaney, Eric; Barkalifa, Ronit et al. (2020): Non-invasive monitoring of pharmacodynamics during the skin wound healing process using multimodal optical microscopy. In BMJ Open Diabetes Research & Care 8 (1), e000974. DOI: 10.1136/bmjdrc-2019-000974

